
Overview
In a year, build a data reporting dashboard for medical device companies to be able to report to the FDA their processes, what’s working well, and where they can improve.
My Role
User Experience Designer
Stakeholders
- Food and Drug Administration
- ISACA Appraisers
- Medical Device Companies
Tools
- Mirro
- InVision
- Illustrator
- Teams
- Outlook
- Figma
- Jira
- GitHub
Research
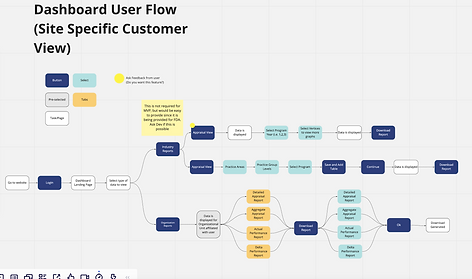
Using the 5 E's Experience model, I evaluated their current user flow by interviewing employees, appraisers and FDA users. Using those insights, I identified areas of opportunity, proposed an improved journey that addressed their needs based on the user, and identified where their data reporting dashboard would be the most useful.
I lead my team in conducting research to find experiences solved similar user needs. Using "How Might We" statements, I collaborated with the Development team, Stakeholders, and my team to identify the "must haves" for MVP, how we might be able to incorporate "nice to have" and phase two features.


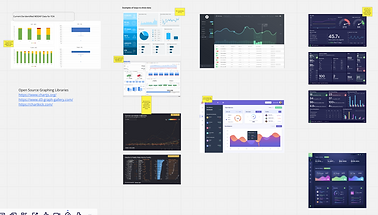

Sketch
Using the How Might We statements as our guide, I lead my team in sketching exercises to rapidly prototype the best solutions to solve our users' needs. We interviewed our users to get feedback on our sketches and incorporated their needs into our the following iterations.

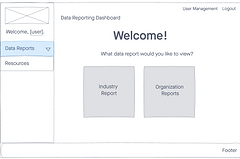


Wireframe
I lead my team in creating basic wireframes to use as a modality to test functionality with users. We were able to simplify the experience as well as test the graphs and design the data in a way is intuitive with FDA users.

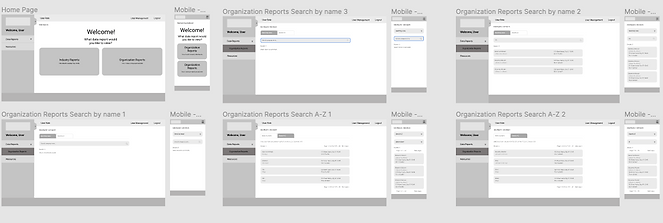

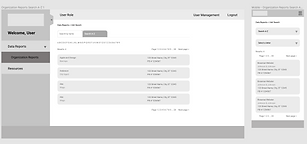
Prototype
At this phase, I lead my team in building out a clickable prototype that we could once again test with users. This prototype served as the final iteration that our Developers used as a guide to build the data reporting dashboard. It was also used as a marketing tool to recruit Medical Device Companies to enroll in this FDA program.
My Learnings
- Getting the development team involved early will help set better expectations for what can be done.
- Lean on user feedback to advocate for your users when speaking with Stakeholders.
What's next?
- Work with the Development team as they build out the data reporting dashboard
- Plan phase 2 to implement more of the "nice to haves" while gathering and incorporating new user feedback.
- Streamline and consolidate the identified tasks to create a "One stop shop" on the reporting dashboard for all users.
Something cool!
"To build a mobile view or not, that is the question!"
They argued that we didn’t need a mobile view for the data reporting dashboard. “No one currently views this type of data in mobile, why would we need it now?” I explained to our stakeholders that users wouldn’t utilize a mobile view if it wasn’t available. After interviewing the FDA individuals that would be using the dashboard, they pointed out how much easier it would be to access the dashboard on mobile, and that they wouldn’t have to wait to get back to the office to check information they needed on hand. It worked! The stakeholders agreed to let us design and build a mobile view after hearing that feedback.


